After China tightened regulation over vaping, People's Daily publishes commentary.
And an original investigation into industry leader RLX Technology
For the sake of simplicity, the terms vaping, e-cigarettes, and ENDS (electronic nicotine-delivery systems) are used interchangeably in this newsletter.
This newsletter is composed of three parts. The first part summarizes China’s recent regulatory development on e-cigarettes, a significant victory for the tobacco control cause.
The second part is a translation of 以强监管规制电子烟 Regulate E-cigarettes with Strong Supervision, published today April 8, 2022, in the People’s Daily, the flagship newspaper of the Communist Party of China.
The third part is an original investigation by Pekingnology into some dubious marketing by 雾芯科技 RLX Technology, China’s e-cigarettes leader listed in New York (NYSE: RLX), including distortion of a World Health Organization (WHO) research displayed in the homepage of RLX Technology’s web site.
In November 2021, the WHO Country Office in China abruptly underlined its stance on e-cigarettes after dubious reports emerged in China purporting to show that WHO had begun to back e-cigarettes.
Pekingnology can report that at least one dubious report in the Chinese media was sponsored by RLX Technology.
After an inquiry from Pekingnology, the RLX Technology-sponsored page vanished from the Chinese media.
There are other dubious reports based on distorted science info on the Chinese-language web, which haven’t disclosed sponsorship and masquerade as news reports to readers.
The brand name of RLX Technology products in China is 悦刻 Yue Ke, by the way.
Since I’m naming a publicly-traded company, let me once again emphasize this newsletter reflects only personal views.
***
Part I
On March 11, 2022, China’s 国家烟草专卖局 State Tobacco Monopoly Administration (STMA) published 电子烟管理办法 [Chinese] Administrative Measures for E-cigarette Management [Unofficial translation via Tobacco Control Laws], which will take effect on May 1.
Earlier, the STMA released a draft [Chinese] of the regulation and its explanations [Chinese] on the draft on December 1, 2021.
It turns out the regulation, compared with the draft, significantly strengthened requirements and imposed many burdens on the manufacturing, advertisements, and sales of e-cigarettes.
The regulation brings strict control, i.e. widespread bans, over advertisements for e-cigarettes under the framework of traditional tobacco
Article 21 The provisions on tobacco advertising in relevant laws, regulations, and rules shall apply to the supervision and management of e-cigarette advertising. Exhibitions, forums and expos promoting e-cigarette products in various forms are prohibited.
The regulation, similar to the recent federal move [NBC News] in the United States, bans flavored e-cigarettes
Article 26 Sale of flavored e-cigarettes other than tobacco flavored ones or e-cigarette products that users can add their own e-vapor matter is prohibited.
Vaping products have to undergo premarket technical reviews [National Cancer Institute] at FDA in the U.S. The Chinese regulation now imposes similar burdens:
Article 19 Electronic cigarette products that have not passed the technical review shall not be sold in the market.
The regulation imposes strict licensing requirements throughout the chain of the e-cigarettes industry
Article 8 The establishment of e-cigarette manufacturers (including production and processing enterprises, brand-holding companies), e-vapor matter manufacturers, and manufacturers of nicotine for e-cigarettes must be reported and approved by the State Council's tobacco monopoly authority before the projects are approved for establishment in accordance with relevant state regulations.
The establishment of the above-mentioned enterprises must be approved by the State Council’s tobacco monopoly authority; they must also obtain the tobacco monopoly manufacturers license and be approved and registered by the market supervision and management authority.
Their separation, merger, and withdrawal must be approved by the State Council’s tobacco monopoly authority, and by the market supervision and management department for the relevant registration procedures.
Their separation, merger and withdrawal must be approved by the State Council’s tobacco monopoly authority and registered with the market supervision and management authority.
Without the tobacco monopoly manufacturer license, the market supervision and management authority shall not approve the application for registration.
No vending machine for e-cigarettes
Article 23 Selling e-cigarette products from a vending machine is prohibited.
The entire e-cigarettes industry can only run in government-designated channels, including their retailing, likely increasing the difficulty to reach consumers.
Article 19 The State Council’s tobacco monopoly authority shall establish a nationwide unified e-cigarette transaction management platform. Manufacturers of e-cigarettes, e-vapor matter, and nicotine used for e-cigarettes, e-cigarette wholesalers, retail entities with a tobacco monopoly license should conduct trading activities through the e-cigarette transaction management platform
Article 23 Any individuals, legal persons, or other organizations should not sell electronic cigarette products, e-vapor matter, and nicotine used for e-cigarettes through information networks other than the e-cigarette transaction management platform provided by these Measures.
The regulation requires prior government approval for e-cigarettes to seek public listings, likely increasing the difficulty of start-ups in this sector to raise funds.
The above-mentioned enterprises should report to, be reviewed, and approved by the State Council’s tobacco monopoly authority prior to their initial public offering of stock shares and being listed.
To be fair, it should also be noted that the strict regulation in effect sets very high entry barriers for existing market entities if the latter successfully resolve compliance issues and remain in the market.
Overall, this is a significant victory for tobacco control for China and also the world.
On e-cigarettes, The FDA says
To date, no e-cigarette has been approved as a cessation device or authorized to make a modified risk claim, and more research is needed to understand the potential risks and benefits these products may offer adults who use tobacco products.
Due to what has been called an ‘epidemic’ of youth use of these products, FDA has prioritized prevention efforts.
FDA’s award-winning public education campaign, “The Real Cost,” continues to prevent youth from tobacco initiation and use. In 2017, the campaign began prioritizing e-cigarette prevention messaging to combat increasing youth vaping rates. “The Real Cost” campaign also educates teens on the health consequences of smoking cigarettes.
The World Health Organization says
Are e-cigarettes and other vaping products dangerous?
Evidence reveals that these products are harmful to health and are not safe. However, it is too early to provide a clear answer on the long-term impact of using them or being exposed to them. They are particularly risky when used by children and adolescents.
Do e-cigarettes (ENDS) cause lung injuries?
There is growing evidence that ENDS could be associated with lung injuries and in recent times e-cigarette and vaping have been linked to an outbreak of lung injury in the USA.
Are e-cigarettes more or less dangerous than conventional tobacco cigarettes? It is difficult to generalize on the risk to health of ENDS as compared with cigarettes or other tobacco products, as this is contingent on a range of factors. Both tobacco products and ENDS pose risks to health. The safest approach is not to use either.
China is the largest producer and consumer of tobacco in the world. There are more than 300 million smokers in China, nearly one-third of the world's total. More than half of adult men are current tobacco smokers. About one in every three cigarettes smoked in the world is smoked in China.
In recent years, vaping has taken off in China. A 2021 year-end report [Chinese] from iResearch estimates that 1.3% of the Chinese population - over 14 million, that is - has taken up e-cigarettes.
Vaping has risen to a global issue and has rightfully been meticulously addressed by responsible news reports and analyses. Here are some from The New York Times in the last several years
The Loophole That’s Fueling a Return to Teenage Vaping. Sales are rising of flavored e-cigarettes using synthetic nicotine that evades regulatory oversight, a gap that lawmakers are now trying to close.
Vaping Is Risky. Why Is the F.D.A. Authorizing E-Cigarettes? The agency has taken a controversial stand on vaping as a way to quit tobacco. This is what the research shows.
Juul Wanted to Disrupt Big Tobacco. Instead It Created an Epidemic of Addiction.
Part II
The People’s Daily, the flagship newspaper of the Communist Party of China, today April 8, 2022, published a commentary on e-cigarettes. Pekingnology hereby offers a full translation. The writer 李思辉 [Chinese] Li Sihui appears to be a current affairs commentator, based on Internet records (I don’t know for sure).
以强监管规制电子烟(人民时评)
(People’s Commentary) Regulate E-cigarettes with Strong SupervisionBy 李思辉 Li Sihui
《 人民日报 》( 2022年04月08日 第 05 版)
Page 5, People’s Daily, April 8, 2022
“A fantastic gadget for quitting smoking”, “Steams of health”, “Trendy technology”... In recent years, some merchants have claimed that e-cigarettes have the effect of helping people quit smoking, improve their health, etc., attracting a large number of consumers. E-cigarette manufacturers have expanded their investment, and e-cigarette stores, authorized resellers, and retailers have sprung up in the fiercely competitive market. As a result, a small number of teenagers have begun to use e-cigarettes, raising widespread concern.
Vaping, like smoking, is harmful to health. As the number of users grows, it has been found that those who tried vaping to quit smoking are prone to using real cigarettes after a period of time. The effect of quitting smoking via vaping is not good. Moreover, relevant agencies noted that e-cigarettes contain nicotine, an addictive substance, and that vaping may cause a host of diseases such as acute lung injury and asthma.
There is growing knowledge among the people that e-cigarettes are essentially cigarettes, and that inhaling will injure the human body. If they are cigarettes, then they should be controlled, and they should not be allowed to slip through supervision under the pretense of “technological products”, following which they will mislead consumers and harm human health.
At the beginning of this year, four government departments including the Ministry of Public Security launched a joint special campaign to ban the sale of e-cigarettes to minors and harshly crack down on crimes involving e-cigarettes in accordance with the law, in an effort to protect the physical and mental health of minors.
Not long ago, the State Tobacco Monopoly Administration issued the Administrative Measures for Electronic Cigarettes, which bans the sale of flavored e-cigarettes other than tobacco flavors cigarettes as well as e-cigarettes to which users can add atomized substances, and prohibits the sale of e-cigarette products to minors as from May 1 this year.
The document clearly stipulates the definition of e-cigarettes and the objects falling under supervision, requires government licensing for the market entities engaged in e-cigarette production, wholesale, and retail, as well as designated channels for sales of e-cigarettes, and so on. Establishing laws and regulations that include e-cigarettes in the scope of tobacco management for effective supervision will certainly contribute to the sound development of the industry and better safeguard the legitimate rights and interests of consumers, particularly minors.
"The letters of the law alone are not enough." [That’s a Chinese proverb.] The soon-to-be-implemented new regulation on the administration of e-cigarettes is targeted and highly feasible. The key lies in truly implementing the rules and regulations in the future.Relevant departments shall work diligently to tighten supervision in accordance with regulation, the industry, and the market entities shall translate the letters in the regulations into thorough, detailed implementation, and the media, schools, and families shall work to raise public awareness of the harms that e-cigarettes may bring ... Only through multi-party cooperation, diverse measures, persistent efforts, and fine governance can we give full play to the regulation and reduce the harm caused by e-cigarettes.
More people will have a rational understanding of e-cigarettes and will develop the habit of consciously smoking less or even quitting smoking if the e-cigarette market is regulated and the e-cigarette industry is overseen. This helps to cultivate a good lifestyle and provide a good social environment for the teenagers’ growing up.
Tobacco control concerns not only the health of young people but also the future of the country and the nation. The outline of the “Healthy China 2030” plan proposes that: “By 2030, the smoking rate for people over 15 years of age will be reduced to 20%.”
In recent years, our country has promoted the fulfillment of the WHO Framework Convention on Tobacco Control across the board, stepped up efforts to control tobacco, leveraged pricing, taxation, law, and other means for more effective tobacco control, conducted in-depth publicity and education on tobacco control, promoted the building of a tobacco-free environment, and strengthened the oversight and law enforcement of tobacco control in public places. Remarkable results have been made in these respects. Thanks to the increasingly detailed measures, we are bound to reduce the public use of e-cigarettes, especially among minors, and contribute to building a healthy China.
Part III
The WeChat blog of the WHO on November 5, 2021, published an article [Chinese] entitled 世卫组织新报告划重点:电子烟是有害的,且尚无充分证据证明其可以作为戒烟工具 New WHO report highlights: e-cigarettes are harmful and there is insufficient evidence to prove they can be used as a smoking cessation tool
But the WHO report on the global tobacco epidemic, 2021: addressing new and emerging products wasn’t new at all - it was already published in August. So why did the WHO - more specifically, its Country Office in China - apparently feel the need to highlight its message?
WHO Country Office in China declined to elaborate when reached by Pekingnology in mid-November 2021.
Around the beginning of November, some “news reports” surfaced on some Chinese media and “self-media” (those not associated with legacy media), claiming new WHO research had proved that “e-cigarettes can help smokers quit smoking has positive public health impact”
https://www.163.com/dy/article/GNNUJVM8051986UM.html
https://www.sznews.com/news/content/mb/2021-11/02/content_24700535.htm
The content in these pieces is identical and most of them did not identify their origin.
Except one.
At the end of this web page, there is a disclosure
以上图为悦刻,授权中国网财经使用
The pictures above are from 悦刻 Yue Ke, and have been authorized to the finance section of China.com for use.
Pekingnology reached out to RLX Technology in mid-November 2021 and the web page http://finance.china.com.cn/roll/20211109/5688813.shtml is now dead. However, an archived page showing the disclosure remains available at https://archive.ph/PHqKU
Many dubious points exist in the RLX Technology-sponsored piece. For example
自今年10月美国食品药品监督管理局(FDA)首次授权电子烟在美国合法销售以来,越来越多的国家开始认可电子烟,调整监管政策,推进本国电子烟的合法化、规范化发展。
Since October this year, after the U.S. Food and Drug Administration (FDA) for the first time authorized the legal sale of e-cigarettes in the United States, more and more countries began to recognize e-cigarettes, adjusting regulatory policies to promote the legalization and standardization of the development of electronic cigarettes in their countries.
10月12日,FDA首次通过了3款电子烟产品的PMTA(Premarket Tobacco Application,烟草上市前申请)审核,因“有数据证明,这3款产品更可能被烟民用来降低其受到烟草伤害的风险”。也就是说,经过层层检测后,FDA认为这3款电子烟产品能帮烟民减害。
On October 12, the FDA passed the PMTA (Premarket Tobacco Application) review of three e-cigarette products for the first time, because "there are data that demonstrate that these three products are more likely to be used by smokers to reduce their risk of harm from tobacco". In other words, after layers of testing, the FDA believes that these three e-cigarette products can help smokers reduce harm.
Basically, the RLX Technology-sponsored piece cited the FDA approval of three specific Premarket Tobacco Applications as examples to insinuate that FDA backs e-cigarettes in general, which is factually incorrect.
The FDA press release said
The manufacturer’s data demonstrates its tobacco-flavored products could benefit addicted adult smokers who switch to these products,
And that simply cannot be applied to the Chinese market where RelxTech and its peers, at the time, market a vast number of flavored e-cigarettes.
To repeat, the disclosure indicates that RLX Technology sponsored that one particular piece with dubious claims, but Pekingnology does NOT have evidence that similar content on other Chinese media was also sponsored by RLX Technology.
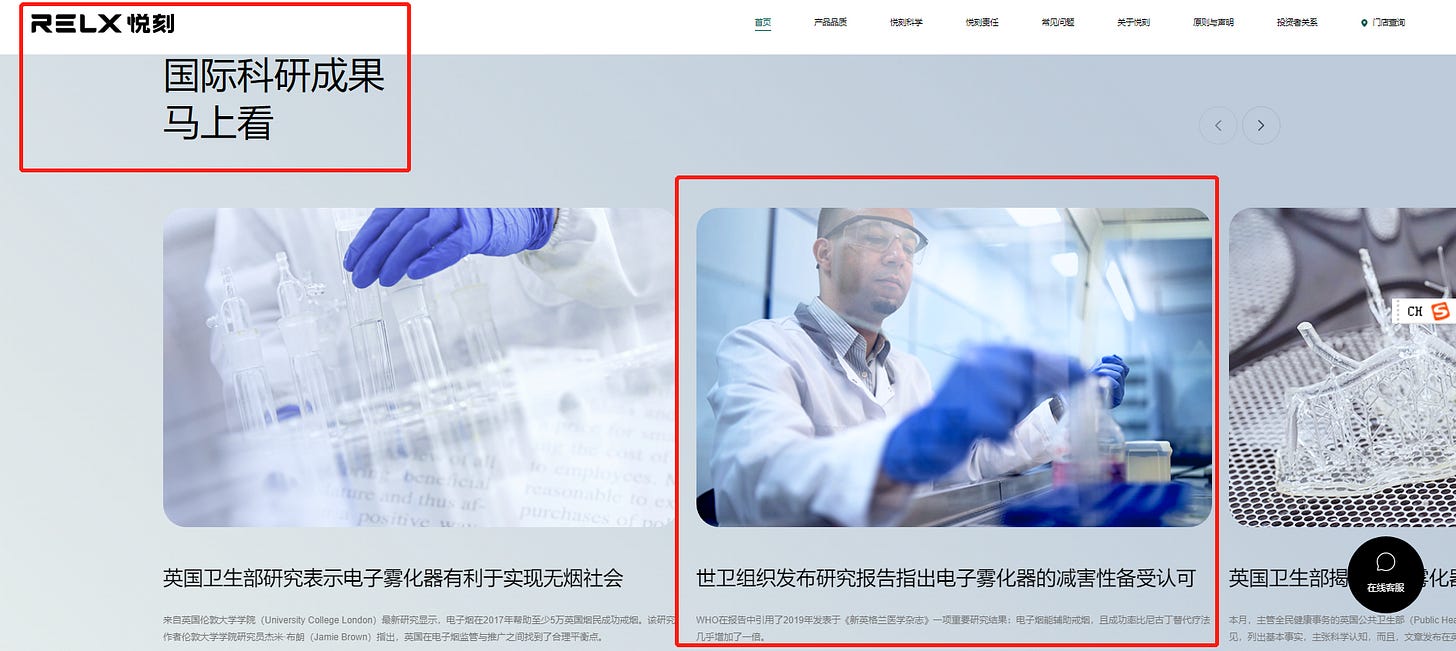
Moreover, RLX Technology’s own website - more precisely its home page - still features a distortion of WHO research.
Published on Feb 28, 2022, which is three months after WHO Country Office in China came out to underline [Chinese] e-cigarettes are harmful and there is insufficient evidence to prove they can be used as a smoking cessation tool, RLX Technology claims in the title
(Interestingly, I have one friend in Brussels and another in the U.S. reporting they can’t access the particular RLX Technology page https://www.relxtech.com/news/17)
世卫组织发布研究报告指出电子雾化器的减害性备受认可
WHO released a study pointing out that the harm reduction of electronic nebulizer is highly recognized
and purports to quote a WHO report in 2020 declaring so.
The problem is that the RLX Technology piece just quoted two of the numerous researches cited in the 317-page 2020 WHO report, and then equated the cherry-picked quotes to the conclusion of 2020 WHO report.
RLX Technology says
健康影响:肯定电子烟减害性
Health effects: affirming the harm-reducing nature of e-cigarettes
“完全使用电子烟代替香烟,可减少使用者接触香烟燃烧产生的多种有毒物和致癌物的机会”。
"The complete use of e-cigarettes in place of cigarettes reduces the user's exposure to the many toxic and carcinogenic substances produced by cigarette combustion."
并且,还有大量证据表明:
And, there is also substantial evidence that.
“使用电子烟会导致尼古丁依赖,但依赖程度低于香烟。”
"Use of e-cigarettes can lead to nicotine dependence, but at a lower level of dependence than cigarettes."
电子烟确有戒烟功效
E-cigarettes do have a smoking cessation effect
WHO也认为,目前不能得出使用电子烟会导致吸烟率增加的结论。 有实验结果表明年轻人使用电子烟后会增加香烟使用可能性,但WHO指出,并无可信数据能够证明这种关联存在因果关系。
WHO also believes that it cannot conclude that the use of e-cigarettes will lead to an increase in smoking rates. There are experimental results that show young people using e-cigarettes will increase the likelihood of cigarette use, but WHO notes that there is no credible data to prove that there is a causal relationship for this association.
今天,依然有许多面目狰狞的偏见,
在忽略事实真相、漠视权威研究。
理性认知电子烟,尤其需要我们放下偏见,
回归独立思考。
Today, there are still many ugly biases that
in ignoring the truth and disregarding authoritative research.
Rational perception of e-cigarettes, in particular, requires us to put aside our biases and
return to independent thinking.
That is a far cry from the 2020 WHO report’s conclusion on ENDS (electronic nicotine-delivery systems), on Page 164
ENDS use by previously nicotine-naïve individuals is inconsistent with public health goals.
Flavoured ENDS liquids contribute to initiation and maintenance of ENDS use among previously nicotine-naïve individuals. They may also be attractive for smokers who want to quit cigarettes.
Under certain circumstances, such as in the context of intensive be havioural counselling, ENDS that deliver nicotine effectively might help some smokers to quit combustible smoking, with positive public health effects. Most of these individuals, however, continue to use ENDS, with uncertain individual health consequences and thus an uncertain public health impact.
Most ENDS users do not quit smoking combustible cigarettes but rather use both ENDS and combustible cigarettes, which, at the least, maintains the substantial health risks associated with cigarette smoking and may increase their health risks.
A representative of RLX Technology said the company has always been consistent in complying with and supporting Chinese regulations, without addressing the specific issues raised by Pekingnology.
(The cover picture comes from Reuters.)





The US FDA, while authorising sale of certain e-cigarette brands, stated: For these products, the FDA determined that the potential benefit to smokers who switch completely or significantly reduce their cigarette use, would outweigh the risk to youth. Link: https://bit.ly/3JpbNS4
This does not appear to be an anti-vaping stance, even though the agency has taken a dim view of flavoured e-liquids (though please keep in mind that 'tobacco' is also a flavour, and so is mint which the agency has allowed). E-cigarettes are now allowed for sale in over 70 nations, which include the entire western countries, with some among them led by the UK & NZ actively encouraging smokers to switch with the aim of bringing down smoking-related mortality and morbidity.
In the context of China which has the largest number of smokers in the world, a shift towards lower-risk smoking alternatives can have major positive public health impact, for which the Chinese tobacco monopoly must adapt to the new environment instead of seeking to block out innovations to protect its massive cigarette sales.
Ofcourse e-cigarette regulation is a must, but its overall aim should be to prevent teen uptake and encourage current smokers to switch, rather than to obliterate the vaping market such that cigarette use and its associated harms are perpetuated. Given that majority of the world's e-cigarettes are produced in China, how well or poorly the country regulates its e-cigarette industry will also have global impact on dealing the tobacco crisis which claims 8 mn lives every year.
Finally, the claim made in People's Daily that "e-cigarettes are essentially cigarettes" is too simplistic to be accurate as vaping is significantly less harmful than smoking which it is meant to replace. Would anyone, for instance, claim greener cars are also cars and therefore should be subject to the same barriers as polluting cars?
The US FDA, while authorising sale of certain e-cigarette brands, stated: For these products, the FDA determined that the potential benefit to smokers who switch completely or significantly reduce their cigarette use, would outweigh the risk to youth. Link: https://bit.ly/3JpbNS4
This does not appear to be an anti-vaping stance, even though the agency has taken a dim view of flavoured e-liquids (though please keep in mind that 'tobacco' is also a flavour, and so is mint which the agency has allowed). E-cigarettes are now allowed for sale in over 70 nations, which include the entire western countries, with some among them led by the UK & NZ actively encouraging smokers to switch with the aim of bringing down smoking-related mortality and morbidity.
In the context of China which has the largest number of smokers in the world, a shift towards lower-risk smoking alternatives can have major positive public health impact, for which the Chinese tobacco monopoly must adapt to the new environment instead of seeking to block out innovations to protect its massive cigarette sales.
Ofcourse e-cigarette regulation is a must, but its overall aim should be to prevent teen uptake and encourage current smokers to switch, rather than to obliterate the vaping market such that cigarette use and its associated harms are perpetuated. Given that majority of the world's e-cigarettes are produced in China, how well or poorly the country regulates its e-cigarette industry will also have global impact on dealing the tobacco crisis which claims 8 mn lives every year.
Finally, the claim made in People's Daily that "e-cigarettes are essentially cigarettes" is too simplistic to be accurate as vaping is significantly less harmful than smoking which it is meant to replace. Would anyone, for instance, claim greener cars are also cars and therefore should be subject to the same barriers as polluting cars?